
BRAINMODEL: precision medicine for brain disorders

A team of researchers from six Dutch knowledge institutions (including coordinator Vrije Universiteit Amsterdam) is introducing new research methods to improve treatment for developmental disorders of the brain. The scientific team will work together with societal partners, clinical associations and companies as the BRAINMODEL consortium and will implement new stem cell-based methods to improve diagnostics and therapy decisions and to develop new therapies. The Netherlands Organisation for Health Research and Development (ZonMw) has allocated a grant of 4 million euros for this work.
For many brain disorders, it is becoming increasingly clear which DNA variation contributes to disease. However, an effective systematic approach for linking this knowledge and the underlying biological mechanisms to treatment options is mostly lacking.
Living nerve cells in a dish
BRAINMODEL’s new approach is based on patient-derived cells with which networks of living nerve cells are produced in a culture dish. These closely resemble the neural networks in our brains. This is known as ‘pluripotent stem cell technology (iPSC)’ and offers new opportunities in terms of understanding human diseases and finding personalized treatments. IPSC-based strategies are particularly promising for developmental brain disorders, because they overcome the problem of not having access to brain tissue to study underlying causes.
Optimal treatment
The BRAINMODEL consortium aims to integrate these methods with diagnostic techniques and relate them to other technologies such as EEG (with which brain activity is measured electronically) to find the best possible treatment for individual patients. BRAINMODEL thus hopes to help optimize therapy choice and make the treatment of brain disorders more successful. At the same time, the researchers will address the ethical and social questions associated with using these technologies together with patients, patient organizations, clinicians, representatives from industry and other societal partners in the consortium.
Linking analyses to measures of outcome
BRAINMODEL’s main objective is to use iPSC-based analyses to detect abnormal cellular functions in material from patients with brain disorders and to link these to clinical outcome measures, such as EEG biomarkers and patient-relevant symptom scales. The latter work will be carried out in collaboration with two clinical centres: the N=You Kenniscentrum (N=You Knowledge Centre) in Amsterdam and the Radboudumc expert center for rare genetic disorders in Nijmegen. The researchers will subsequently test whether abnormal cellular properties, which cause an imbalance between the positive and negative signals that nerve cells send to one another, can be remedied with existing medication in the iPSC-based analyses. If this is the case, the physicians at the associated clinics can use this medication to treat the patients in question. If no existing medication can be found to remedy the abnormal properties, BRAINMODEL will use specially-developed nanochip technology to carry out iPSC-based analyses on a larger scale. This method will enable large-scale drug research, including the design of new drugs. To this end, cellular analyses will be automated and scaled up by culturing human nerve cells on nanochips. This way, a hundred times more drugs can be tested and new drugs can be developed.
PSIDER
The BRAINMODEL consortium was founded by Matthijs Verhage (Vrije Universiteit Amsterdam and Amsterdam UMC, chairperson) and Nael Nadif Kasri (Radboudumc, vice chairperson). Other knowledge institutions represented in BRAINMODEL are Erasmus MC, University of Twente, Radboud University and the Radboudumc expert center for rare genetic disorders. The consortium has received a grant of four million euros from ZonMw within the framework of the grant call ‘Multidisciplinary Consortia Programme Pluripotent Stem cells for Inherited Diseases and Embryonic Research’ (PSIDER). For more information and a list of all the partners in the consortium, please see Consortium.

BRAINmodel open day 2026
Geslaagde open dag voor STXBP1- en SYT1-patiëntenfamilies
Op zaterdag 9 mei openden we de deuren van het VU Onderzoeksgebouw voor een open dag voor families van patiënten met de zeldzame STXBP1- en SYT1-gerelateerde aandoeningen.
Onderzoekers van het BRAINMODEL consortium, werkzaam op de afdeling Functionele Genoomanalyse (CNCR), gaven de bezoekers een unieke inzage in hun werk. Het programma startte met een presentatiesessie waarin onderzoekers Jan Sprengers, Niels Cornelisse, Noortje van Geest en Claudia Persoon de huidige stand van het onderzoek aan STXBP1- en SYT1-gerelateerde aandoeningen toelichtten. Er werd uitgelegd hoe patiënt-eigen iPSC-derived neuronen worden gebruikt om de ziektemechanismen beter te begrijpen en potentiële medicijnen te testen. Aansluitend kregen de families een rondleiding door de laboratoria en een demonstratie van de elektrofysiologie- en microscopietechieken die gebruikt worden op de afdeling.
De open dag zorgde voor waardevolle interactie en onderling begrip tussen de wetenschappers en patiëntfamilies. We kijken terug op een zeer geslaagde dag!
Successful open day for STXBP1- and SYT1-patient families
On Saturday May 9th, we opened the doors of the VU Research Building for an open day for families of patients with rare STXBP1- and SYT1-related disorders.
Researchers from the BRAINMODEL consortium, working at the department of Functional Genomics (CNCR), provided the visitors with a unique insight into their work. The program started with a presentation session in which researchers Jan Sprengers, Niels Cornelisse, Noortje van Geest, and Claudia Persoon explained the current state of research on STXBP1- and SYT1-related disorders. They explained how patient-own iPSC-derived neurons are used to better understand disease mechanisms and test potential candidate drugs. Afterwards, the families attended a tour through the laboratories, with demonstrations of the electrophysiology and microscopy techniques used in the department.
The open day sparked valuable conversation and mutual understanding between the scientists and patient families. We look back on a highly successful day!

Million Dollar Bike ride for research of STXBP1-related disorders
On Saturday June 8, 2024, our teams successfully biked the European Million Dollar Bike Ride, a fundraising event to raise money for research into STXBP1-related disorders! Together they raised more than 12.000 euros.
The teams consisted of researchers and clinicians from the Functional Genomics department of Vrije Universiteit Amsterdam, Human Genetics department of Amsterdam UMC, Amsterdam Neuroscience, Radboud UMC, University of Antwerp, and Antwerp University Hospital, as well as family members of STXBP1 patients. Each team biked more than eighty kilometers to the mid-point in Dordrecht, halfway between the labs of Amsterdam and the labs of Antwerp. In Dordrecht, all bikers were welcomed by supporters, family members of patients and colleagues.
We would like to thank all cyclists for their great achievement, the supporters for their enthusiasm and the sponsors for their donations. Especially Amsterdam Neuroscience for their generous donation of € 2.000,00! It has been heartwarming to see the overwhelming support for our cause. In total we raised € 6.359,00 and Penn State University will double this amount.
All donations have been transferred to the Million Dollar bike ride organization in the United States (team STXBP1). The Penn Medicine Orphan Disease Center will distribute the funds to the international scientific community through grant proposal applications. They will monitor and manage progress of the science and spending of the funds.
See the MDBR website for more information.
Million Dollar Bike Ride 2024: BRAINmodel researchers race for rare disease research
Researchers from the BRAINmodel consortium and colleagues from Belgium are preparing to participate in the European satellite event of the 2024 Million Dollar Bike Ride. The ride, orchestrated by the Orphan Disease Center in Philadelphia, aims to generate funds earmarked for critical rare disease research grants, with our teams’ funds specifically dedicated to STXBP1 studies.
Scheduled for Saturday, June 8th, the race will see both teams embarking on an 80-kilometre journey to a designated midpoint between Antwerp and Amsterdam.
Building upon the success of the 2022 event, where over 10,000 euros were raised, we aspire to make this year’s race an even greater triumph.
Your support is paramount in our quest. Kindly consider contributing through the following link:
https://www.gofundme.com/f/million-dollar-bike-ride-for-stxbp1-patients-2024
Let’s race towards a brighter future for STXBP1 patients together!

Kleefstra Syndroom Dag – 29 september 2023 in Nijmegen
Op 29 september van 13.30-17.30 vindt er een Kleefstra Syndroom dag plaats voor families en patiënten, georganiseerd door het Radboud UMC en ZeldSamen. Het programma bestaat uit verschillende sprekers, onder andere van het BRAINmodel consortium, en vindt plaatst op het Radboud UMC. Zie onderstaande flyer voor meer informatie over het programma.
U kan zich hier aanmelden.

STXBP1 informatieavond 21 juni 2023

Op woensdag 21 juni aanstaande organiseert het STXBP1 team (VU Amsterdam & Amsterdam UMC) een STXBP1 informatieavond voor verzorgers van STXBP1 patiënten. De sessie zal online te volgen zijn, maar geïnteresseerden zijn – indien gewenst en haalbaar – ook van harte welkom in het Amsterdam UMC, locatie AMC. We verzoeken u zich aan te melden via deze link: https://forms.gle/dQ4mvvsUAq7jV7Me6

Kleefstra Syndrome Scientific Conference 2023
Nederlands hieronder
On June 1th-June 2nd, the European Joint Program together with Prof. Tjitske Kleefstra, is organizing a Kleefstra Syndrome Scientific conference in Ljubljana. This event is open for everyone and free of charge for online participants.
“The Kleefstra Syndrome Scientific Conference 2023: “Moving towards the uptake and use of Artificial Intelligence (AI) in research and clinical work” focuses on Kleefstra syndrome, a rare genetic disorder with app. 1000 diagnosed patients worldwide. Due to the rarity of the disease, a global perspective is needed to foster new research insights. The event brings together Kleefstra syndrome PAOs, clinicians and researchers from different domains having a common interest: share new research findings related to Kleefstra syndrome that will lead the Kleefstra community to optimize current care and to reach their final goal, the discovering of a life-changing treatment and cure for Kleefstra syndrome. A special focus is given to artificial intelligence (AI), which is generally still something new for the rare disease communities, but it can play a crucial role, especially in shortening the time needed for new research insights.” (https://idefine-europe.org/conference/)
Check the website if you are interested!
—————————————————————————————————————————————–
Op 1 t/m 2 juni organiseert het European Joint Programme samen met prof. dr. Tjitske Kleefstra een Kleefstra Syndrome Scientific congres in Ljubljana. Dit evenement is voor iedereen toegankelijk en gratis voor online deelnemers.
“The Kleefstra Syndrome Scientific Conference 2023: “Moving into the uptake and use of Artificial Intelligence (AI) in research and clinical work” richt zich op het Kleefstra-syndroom, een zeldzame genetische aandoening met ongeveer 1000 gediagnosticeerde patiënten wereldwijd. Vanwege de zeldzaamheid van de ziekte is een mondiaal perspectief nodig om nieuwe onderzoeksinzichten te bevorderen. Het evenement brengt clinici en onderzoekers uit verschillende domeinen samen met een gemeenschappelijke interesse: nieuwe onderzoeksresultaten met betrekking tot het Kleefstra-syndroom delen die de Kleefstra-gemeenschap ertoe zullen aanzetten de huidige zorg te optimaliseren en hun uiteindelijke doel te bereiken: het ontdekken van een behandeling en genezing van het Kleefstra syndroom. Er wordt speciale aandacht besteed aan kunstmatige intelligentie (AI), wat een cruciale rol kan gaan spelen, vooral bij het verkorten van de tijd die nodig is voor het genereren van nieuwe onderzoeksinzichten.” (https://idefine-europe.org/conference/)
Check de website als je geïnteresseerd bent!
Clinical research progress within BRAINmodel
Within the BRAINmodel project we aim to link clinical outcome measures in patients with brain disorders to abnormal cellular functions in material (iPSC-derived neurons) from these same patients. To do this, we need to prepare many aspects of the project, and in the last months we have made significant progress which we would like to share with you.
Our research protocol has been approved by the ethics committee and also by the boards of directors of both Amsterdam UMC and Radboudumc! An important step before we could start our study.
At this moment we are recruiting participants for the BRAINmodel study. We are very happy to share that we have already included nine children in our research. We will carefully map out their medical problems, behaviour, metabolism and potential issues with their immune system. We will also perform an EEG recording to measure their brain activity. For a subset of patients we will combine this with research on the patient’s own stem cells, grown into functional neurons within the lab. With this, we hope to gain a better understanding of stimulus processing and the balance between over- and under-stimulation.
Last week we tested our EEG equipment at Radboudumc. And it works! That is why we are now completely ready to meet the first patients for BRAINmodel.
Sietske van Till publishes article in the magazine of patient organization ZeldSamen.
BRAINmodel PhD student Sietske van Till (Erasmus MC) has published an article in the magazine ZeldSamen about her research towards the ethical implications of the use of stem cells to study neurodevelopmental disorders. ZeldSamen is a dutch patient organization offering information, contact and recognition for parents after the diagnosis of a rare genetic syndrome. They collaborate within the BRAINmodel project as stakeholders.
To gain more insight in parents’ overall experiences and their moral perspectives on stem cell research, Sietske van Till and Dr. Eline Bunnik started an interview study among parents of children with the Kleefstra-syndrome or an STXBP1 mutation. In her article, Sietske explains more about the BRAINmodel study, the interviews and how to participate in this research.

Starting 2023, we will initiate the clinical research workpackages within the BRAINmodel project, in which multiple children with SNAREopathies or Chromatinopathies will be investigated, see also our flyer. If you are interested in participating, please contact brainmodel.gen@radboudumc.nl.
Belgian & Dutch STXBP1 labs bike to raise money for research

We bike together to raise money for research towards new treatments for the rare disorder STXBP1 Encephalopathy!
We will raise money for rare disease research on Saturday, June 11, 2022, as European counterpart of The Million Dollar Bike Ride organized by the Orphan Disease Center in the US.
Researchers working on STXBP1 encephalopathy from the VU University and Amsterdam UMC (Netherlands) and University of Antwerp and Antwerp University Hospital (Belgium) will bike towards a halfway point between their labs in Amsterdam and Antwerp. With this combined 170 kilometer bike ride, we hope to raise as much money as possible to aim for a better future for patients suffering from rare disorders. All donations will be transferred to the STXBP1 team of The Million Dollar Bike Ride campaign of the Orphan Disease Center, which supports research towards new treatments for rare disorders.
Please donate here.
If you are interested in riding along with us, or would like to join us at the middle point, please contact us at stxbp1@amsterdamumc.nl.
STXBP1 Encephalopathy
STXBP1 Encephalopathy is a severe genetic disease caused by a disease causing variant in the STXBP1 gene. STXBP1-E patients show a developmental delay in their first years, typically resulting in intellectual disability at a later age. Epilepsy is a common symptom (89% of patients) and is difficult to treat. Many patients also suffer from additional symptoms, such as movement disorders (e.g. muscle hypotonia, ataxia) or neuropsychiatric features such as autism spectrum disorder. The severity of the disorder differs between patients, and until now there is no disease-modulating treatment available.
We, researchers at the Vrije Universiteit (VU) Amsterdam and the Amsterdam UMC, the Netherlands, as well as the VIB Center for Molecular Neurology at the University of Antwerp and Antwerp University Hospital, Belgium, work together to better understand STXBP1- Encephalopathy. We aim to improve diagnosis and therapeutic interventions, and study the disease to understand how mutations in STXBP1 lead to STXBP1 encephalopathy. For more information on the disorder and our research, visit our websites stxbp1.cncr.nl and Sarah Weckhysen lab .
First BRAINmodel paper published
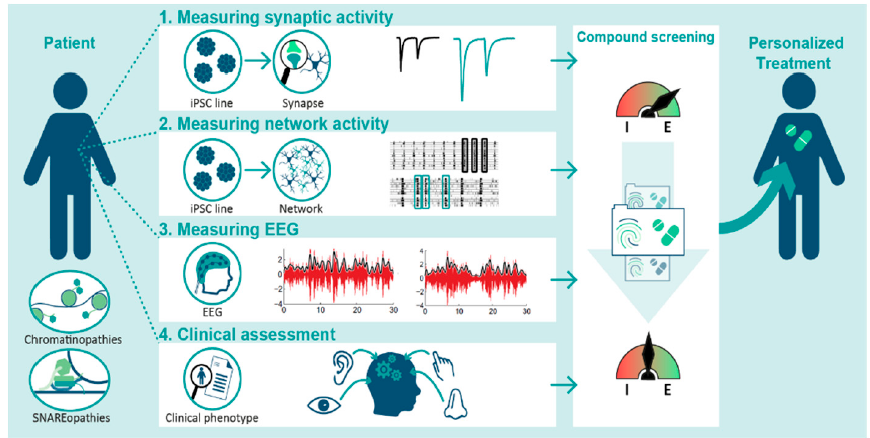
The first results of the BRAINmodel consortium have been published as a concept paper in the scientific magazine Genes. This is a flying start for the consortium, which just started a few months ago, and it is great to see the recognition of our work and plans by the scientific community.
In the paper ‘Following Excitation/Inhibition Ratio Homeostasis from Synapse to EEG in Monogenetic Neurodevelopmental Disorders’ we present our strategy to integrate multiple levels of neuroscientific investigations to develop personalized treatment strategies in patients with neurodevelopmental disorders. We focus on excitation/inhibition ratio homeostasis across cellular and in-patient measurements in patients with chromatin- and SNAREopathies.
Please find the full text: https://doi.org/10.3390/genes13020390






